
The early results showed that, for 24% of patients receiving this new therapeutic combination, no trace of cancerous cells was found in tissues taken during the surgical procedure - compared with just 2% for patients receiving chemotherapy alone.Īt the 2022 AACR Meeting, Prof. With this neoadjuvant treatment combining immunotherapy with chemotherapy, we are now able to significantly reduce this risk of recurrence and improve patient survival 
Although surgery is effective for patients with non-small cell lung cancer, many of them suffer from recurrence. The search for therapeutic options with neoadjuvant (before surgery) or adjuvant (after surgery) treatments is thus vital for improving the outcomes of these patients in the long term.Ĭoordinated since 2017 by teams from the Curie-Montsouris Thorax Institute, the results of the international phase-3 study CheckMate-816 reveal the effectiveness of administrating neoadjuvant treatment combining immunotherapy with chemotherapy in 358 patients suffering from non-metastatic NSCLC.įive years after beginning enrolling patients in this trial, today I am delighted to present the phase-3 results that truly change the outcomes for our patients, based on the work of a team of pulmonologists, surgeons, oncologists and expert pathologists. Many patients with non-metastatic NSCLC are cured by surgery but 30 to 55% of them develop recurrence. Among the two main types of lung cancer, non-small cell lung cancer (NSCLC) accounts for up to 84% of diagnoses, the majority of which (around 60%) are non-metastatic. It is the third most prevalent cancer in France, and its incidence is rising sharply among women. The exploration of these studies showcased the potential for improving patient outcomes and the ongoing evolution in lung cancer therapy.Lung cancer is the leading cause of cancer death throughout the world, with around 1.8 million deaths in 2020. In summary, the discussion reflected on significant advancements in lung cancer treatment, emphasizing the need for additional data to refine treatment choices and further expand the available options for different patient subsets. Reuss highlighted the potential of other compounds, such as patritumab deruxtecan, in the treatment landscape, especially in patients with EGFR mutations, offering more options in this challenging space. The dialogue addressed the need for further investigation and data analysis in these areas. The conversation also touched upon the potential role of dat DxD in patients with actionable mutations who have progressed on upfront TKIs. The results revealed a modest progression-free survival advantage, primarily visible in non-adenocarcinoma subgroups. In metastatic non-small cell lung cancer, the discussion covered the TROPION Lung 01 study, exploring the antibody-drug conjugate dat DxD compared to standard treatment. TROPION Lung 01 study, exploring the antibody-drug conjugate The conversation explored the balance between chemotherapy and alectinib in treatment strategies for these patients. The ALINA trial investigated the role of alectinib in resectable non-small cell lung cancer with ALK-positive mutation, displaying a notable benefit in disease-free survival, particularly in the CNS, which could be groundbreaking for this patient population. ALINA trial investigated the role of alectinib 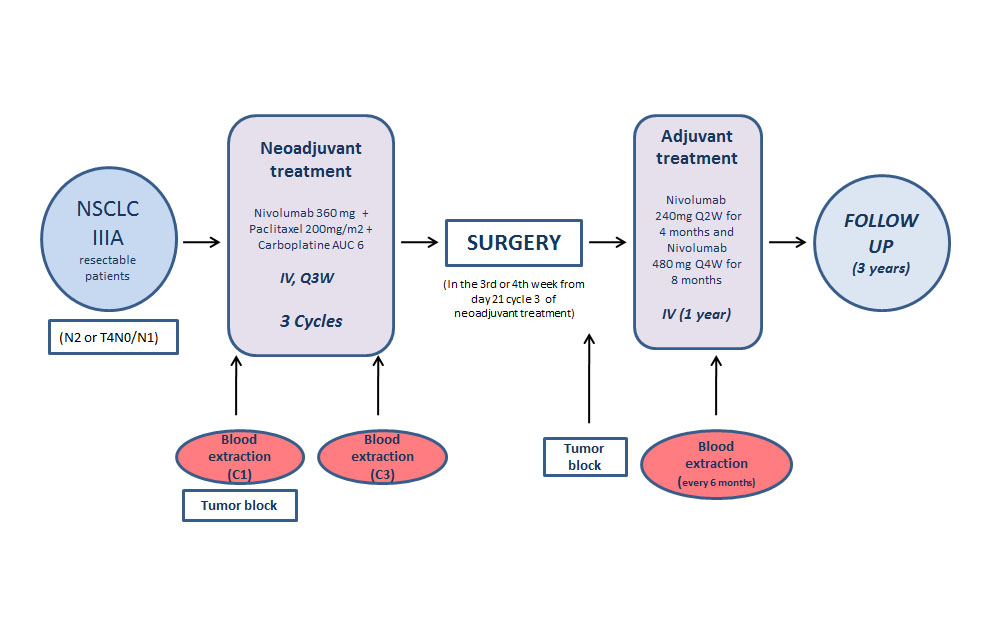
This discussion highlighted the complexities of treatment decisions in this evolving landscape. Regarding the selection between CHECKMATE 816 and KEYNOTE 671 for patients, they emphasized the need for additional data, especially focusing on factors like pathologic response, disease stage, and biomarker analysis. The FDA also approved this regimen for certain patient criteria, marking a significant step in enhancing patient outcomes. 
This benefit was observed across different disease parameters, indicating practice-changing implications. The results showed an overall survival benefit, demonstrating a hazard ratio of 72 in favor of the IO chemo combination. KEYNOTE 671, a phase 3 study, examined the peroperative IO chemo combination and its impact on overall survival. Joshua Reuss from Georgetown Lombardi Kansas Center. They segmented the lung cancer highlights into two key studies: KEYNOTE 671 focusing on resectable non-small cell lung cancer and the Alina trial for ALK-positive patients. Rahul Gosain, MD and Rohit Gosain, MD, referred to as the oncology brothers, reflected on the impressive content presented at ESMO 2023. Top 4 ESMO Trials: KEYNOTE 671, CHECKMATE 816, ALINA, TROPIONĭrs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
